KSL Investigates: What can antibody testing tell us about immunity?
May 4, 2020, 10:25 PM | Updated: Feb 7, 2023, 3:38 pm
SALT LAKE CITY – Scientists around the world are desperately trying to figure out the answer to the question, “who is immune to COVID-19, and can that knowledge help us get life back to normal?”
Many people think the answer lies in antibody testing, but with so many tests now flooding the market, the KSL Investigators wanted to find out if the tests can really be trusted.
Looking For Answers
John Nield is one of many Utahns who wants to know if he’s already contracted COVID-19.
Back in early March, he and his wife took a road trip to California. They hit Monterey, Big Sur and San Luis Obispo.
“We got home, and we got sick the following week,” said Nield. “We had headaches and coughs and achiness.”
Turns out, the area they visited was just south of Santa Clara County, where two Californians died of novel coronavirus in February. Their deaths are now shown to be the country’s earliest.
“That kind of alarmed me and I thought, well maybe I’ve had the coronavirus,” he said.
Nield thinks an antibody test could give him the answers he needs to get back to his regular life.
“I could take my grandkids fishing,” said Nield. “I’d be immune to it, I would think.”
Testing For Antibodies
“One of the secrets to getting life back to normal is figuring out who already has immunity,” said Kenneth Ward.
Ward is the lab director at Predictive Laboratories in Salt Lake City. It’s one of several labs in Utah that has developed an antibody test, and just one of dozens around the country.
“Some will have a better test than others, and we believe we’ll be at that top tier with one of the best tests,” said Ward.
When you’re exposed to a foreign pathogen, like a virus, your body produces antibodies. These proteins latch onto the virus and destroy it, preventing it from making copies of itself and further spreading the infection.
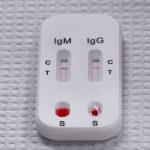
An antibody test looks for antibodies. Their presence most likely means you were exposed to COVID-19.
Predictive Labs has developed a rapid antibody test. With the prick of a finger, they claim you’ll know within 15 minutes whether you’ve developed COVD-19 antibodies – if the test is accurate.
Their test isn’t currently FDA approved, but neither are most antibody tests out there. In fact, out of at least 150 tests on the market, the FDA has verified and authorized just eight so far.
Even those eight only went through an emergency review process where a lab submits all their documentation to the FDA for scientists to look at.
“The FDA process for getting a diagnostic test approved is usually a very long process,” said Ward. “It lasts years.”
Because of the pandemic, the FDA is now giving emergency approval in just days or weeks.
Predictive Labs thinks their approval is coming any time.
“I don’t worry about the companies that are going through the FDA process,” said Ward. “I do worry about the companies who just set up a website and manufacture something in their garage.”
Congress is also concerned. They are sounding the alarm after early data from researchers in California who “tested the tests” showed troublingly high rates of false results. One in three antibody tests they analyzed produced false positives more than 10% of the time.
Not A Free Pass
University of Utah Health is currently doing antibody testing on their frontline medical workers, as well as patients. They use a test from ARUP, one of the eight tests that has FDA approval.
Dr. Emily Spivak is an Associate Professor of Medicine and Infectious Diseases at the U. She said she’s worried any “positive” test, false or not, will give people the wrong idea.
“There’s concern about people seeing it as a free pass, in my opinion, to go back into society, large gatherings and parties and things like we did before,” she said.
The idea of an “immunity passport” has garnered a lot of attention. Many people think it’s the key to reopening the country, but even the World Health Organization said there is just not enough evidence that antibody tests can prove immunity.
“There’s no evidence yet to confirm that the antibody translates into that patient being immune or being protected from another COVID-19 infection,” said Spivak. “I’d caution people that the antibody test probably really should not, at least in the short term, change our practices.”
For now, that means even if folks like Nield can get an antibody test, they’ll still have to wait a little longer for answers.
“I would like to know for my own peace of mind,” said Nield.
Doctors said antibody testing will be very helpful in determining how widespread the virus has been so far, but we could still be months away from figuring out whether having antibodies equals having immunity.
At least two other Utah companies have developed antibody tests as well: Quansys Biosciences and Medsential. They are also waiting on FDA approval.