Trials Underway To See How Well COVID Vaccines Protect Younger Teens
Mar 1, 2021, 1:43 PM
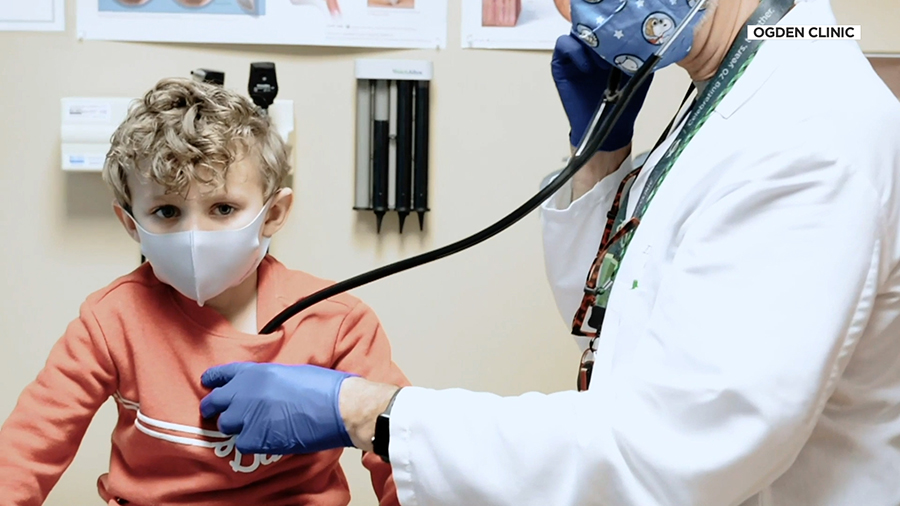
SALT LAKE CITY, Utah – Trials are currently underway to see how well current COVID-19 vaccines work to protect children.
One student in Utah spoke with KSL about why she wanted to get involved in the process.
Few people felt the needle as early as Cadence Eagar. The 17-year-old high school senior got both doses of the vaccine in November. It was part of a Pfizer trial for people 16 and older.
“The whole point of my daughter and I volunteering to do this study was just … to get the vaccine to roll out as fast as possible,” Cadence’s mom said.
Since then, things have picked up. Now, people as young as 16 with certain medical conditions are eligible to receive the vaccine.
Andrew Pavia, chief of pediatric infectious disease at University of Utah Healthcare, said the people least able to fight off the virus are also not very good at responding to vaccines.
He said generally teens at least 16 years old respond about the same as adults.
“We don’t expect there to be safety problems with these vaccines in any of these groups,” Pavia said. “It’s really a question of whether it works as well as we want it to.”
A more intensive study is now underway to see how kids as young as 12 respond.
“I can’t wait for that time because I’d like to see teenagers protected,” Pavia said. “I’d like to see them not be able to spread disease to grandparents.”
He said he hopes to see the FDA sign off on the vaccine for those 12 and older before the next school year.
“When you get down to the young age groups, then things get more complicated,” he said. “You may need to lower the dose.”
Cadence, in the meantime, is continuing to report to Pfizer how she’s doing each week.
“It’s worth it to me,” she said. “Hopefully, everybody can have access to it, and we can all be protected.”